Greenhouse gases: Sources and Sinks
Getting Started
Most greenhouse gases are produced naturally, however their production is increased by anthropogenic factors. Also, there are some manmade greenhouse gases, such as chlorofluorocarbons (CFCs), hydrofluorocarbons (HFCs) and perfluorocarbons (PFCs), as well as sulfur hexafluoride (SF6).
Principal Greenhouse Gases
- carbon dioxide
- water vapour
- methane
- ozone
- nitrous oxide
- CFCs and related manmade greenhouse gases
Carbon Dioxide

Natural sources of carbon dioxide include cellular respiration, volcanic eruption, and the burning of organic matter. Natural sinks of carbon dioxide include living things and oceans. Living things act as sinks as plants capture carbon dioxide and use it as fuel to survive and grow, and are transferred to other organisms because they are at the bottom of the food chain. Oceans also act as sinks. CO2 dissolves in the ocean and some of the CO2 forms calcium carbonate (which sinks to the ocean floor).
Anthropogenic sources of carbon dioxide include deforestation caused by humans, burning fossil fuels for energy (where it is used for transportation, electricity generation, industry purposes), and the respiration of living animals, particularly farmland animals. All of the above causes the release of more greenhouse gases into the atmosphere. While there are many known anthropogenic sources of carbon dioxide, there are also a few anthropogenic sinks of carbon dioxide. Anthropologically constructed carbon dioxide sequestration techniques can help reduce emissions created by anthropogenic sources by capturing and storing carbon dioxide in other forms (like methanol or hydrocarbons).
Anthropogenic sources of carbon dioxide include deforestation caused by humans, burning fossil fuels for energy (where it is used for transportation, electricity generation, industry purposes), and the respiration of living animals, particularly farmland animals. All of the above causes the release of more greenhouse gases into the atmosphere. While there are many known anthropogenic sources of carbon dioxide, there are also a few anthropogenic sinks of carbon dioxide. Anthropologically constructed carbon dioxide sequestration techniques can help reduce emissions created by anthropogenic sources by capturing and storing carbon dioxide in other forms (like methanol or hydrocarbons).
Water Vapour

Water vapour is created all over the world during the water cycle. Water vapour itself traps energy and reflects it back to the earth, fuelling the greenhouse effect. Its sinks are mainly natural, with cold temperatures in general causing condensation and precipitation and plants (some of which derive a part of their water supply during sublimation).
Methane

There is much less methane in the atmosphere than carbon dioxide, but it is approximately 23 times more affective than carbon dioxide at manipulating thermal energy.
Natural sources of methane include biological processes such as plant decomposition in swamps and animal digestion and permafrost in Arctic regions (where the grounds are known to contain high levels of methane gas). Also, wetlands are natural sinks due to the fact that when the water table is high, emissions from soils and plants do not have to pass through layers of soil rich in methanotrophic bacteria. This means that a high water table causes methane produced in soils to diffuse more easily into atmosphere.
Natural sinks of methane include methanotrophs in Earth's soils and some components in Earth's atmosphere. Methanotrophs are organisms that use methane from either the soils or directly from the environment to grow, depleting levels of methane given off by decomposition in Earth's soils. Also, in the atmosphere, components of the atmosphere such as the hydroxyl radical and chlorine gas react with methane to create other compounds, effectively removing it from the atmosphere in that form.
Anthropological sources include livestock, rice agriculture, landfills, burning of biomass, and fossil fuel mining. Livestock raised by humans produce 37 % of anthropogenic methane through livestock flatulence and eructation. Rice agriculture is a source of methane due to the fact that they act as wetlands but they are specifically made by humans for the production of rice. Landfills are another large contributor of methane, basically being a mass amount of decomposition in one area. Methanotrophic bacteria aid in processing some of the methane produced but still 90% of it is released into the atmosphere.
The burning of biomass is a source of methane because of the fact that incomplete burning of organic matter produces methane. Lastly, fossil fuel harvesting is an anthropogenic source of methane due to it being released when it is harvested. Methane is a large component of natural gas, and natural gas can be found in oil reservoirs and in caves where coal is mined. During the processing of these materials, some methane-containing natural gas is released into the atmosphere. There are no anthropogenic sinks of methane.
Natural sources of methane include biological processes such as plant decomposition in swamps and animal digestion and permafrost in Arctic regions (where the grounds are known to contain high levels of methane gas). Also, wetlands are natural sinks due to the fact that when the water table is high, emissions from soils and plants do not have to pass through layers of soil rich in methanotrophic bacteria. This means that a high water table causes methane produced in soils to diffuse more easily into atmosphere.
Natural sinks of methane include methanotrophs in Earth's soils and some components in Earth's atmosphere. Methanotrophs are organisms that use methane from either the soils or directly from the environment to grow, depleting levels of methane given off by decomposition in Earth's soils. Also, in the atmosphere, components of the atmosphere such as the hydroxyl radical and chlorine gas react with methane to create other compounds, effectively removing it from the atmosphere in that form.
Anthropological sources include livestock, rice agriculture, landfills, burning of biomass, and fossil fuel mining. Livestock raised by humans produce 37 % of anthropogenic methane through livestock flatulence and eructation. Rice agriculture is a source of methane due to the fact that they act as wetlands but they are specifically made by humans for the production of rice. Landfills are another large contributor of methane, basically being a mass amount of decomposition in one area. Methanotrophic bacteria aid in processing some of the methane produced but still 90% of it is released into the atmosphere.
The burning of biomass is a source of methane because of the fact that incomplete burning of organic matter produces methane. Lastly, fossil fuel harvesting is an anthropogenic source of methane due to it being released when it is harvested. Methane is a large component of natural gas, and natural gas can be found in oil reservoirs and in caves where coal is mined. During the processing of these materials, some methane-containing natural gas is released into the atmosphere. There are no anthropogenic sinks of methane.
Ozone
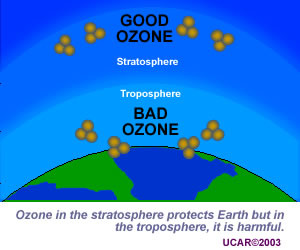
Ozone gas is what makes up ozone layer in stratosphere that helps filter out harmful level of UV radiation. However, in troposphere it only negatively contributes to the greenhouse effect. The atmosphere itself is a natural source of ozone, as ozone is created in the stratosphere naturally due to UV radiation breaking up dioxygen molecules and them coming back together as ozone. Natural sinks of ozone is exactly the same as what creates it: conditions in the atmosphere. Atomic oxygen created by UV radiation introduced to ozone will turn it into two dioxygen molecules.
Ozone with anthropogenic sources remain in the troposphere, mixing with chemicals from man-made pollution to create smog. Incomplete combustion of fossil fuels (e.g. industrial, transportation vehicles, etc.) is an example of an anthropogenic source of ozone. To this date, there are no anthropogenic sinks of ozone.
Ozone with anthropogenic sources remain in the troposphere, mixing with chemicals from man-made pollution to create smog. Incomplete combustion of fossil fuels (e.g. industrial, transportation vehicles, etc.) is an example of an anthropogenic source of ozone. To this date, there are no anthropogenic sinks of ozone.
Nitrous Oxide

Nitrous oxide is almost three hundred times more effective as a greenhouse gas than CO2, but there are much lower concentrations of it found in the atmosphere compared to CO2. Natural sources of nitrous oxide include being a product of the oxidation of ammonia in the atmosphere, and from nitrogen in soils, particularly in tropical zones (created by bacteria in water and soils). Some natural sinks of nitrous oxide include the stratosphere, where nitrous oxide undergoes photolysis due to exposure to UV radiation and becomes N2 and O. It also meets with atomic oxygen and becomes nitric oxide, which is a compound that helps regulate the amount of ozone in the stratosphere.
Anthropogenic sources of nitrous oxide include agriculture, transportation, factories, and fossil fuel use. Agriculture released nitrous oxide into the atmosphere through the use of fertilizers used in the agricultural industry contain nitrogen. Transportation releases nitrous oxide into the air when motor fuels are burned. It is also produced in factories as a by-product of other chemical reactions used to make synthetic materials. Lastly, fossil fuel use (more specifically, the burning of fossil fuels for energy) releases nitrous oxide into the atmosphere as a result of the incomplete combustion that occurs when they are burned for their uses. Like many other greenhouse gases, there are no anthropogenic sinks for nitrous oxide.
Anthropogenic sources of nitrous oxide include agriculture, transportation, factories, and fossil fuel use. Agriculture released nitrous oxide into the atmosphere through the use of fertilizers used in the agricultural industry contain nitrogen. Transportation releases nitrous oxide into the air when motor fuels are burned. It is also produced in factories as a by-product of other chemical reactions used to make synthetic materials. Lastly, fossil fuel use (more specifically, the burning of fossil fuels for energy) releases nitrous oxide into the atmosphere as a result of the incomplete combustion that occurs when they are burned for their uses. Like many other greenhouse gases, there are no anthropogenic sinks for nitrous oxide.
CFCs and Related Manmade Greenhouse Gases

"CFCs and related man-made greenhouse gases" refers to man-made gases consisting of one carbon atom and at least one of the halogens (fluorine, chlorine, iodine, and bromine). Most fall into the category of chlorofluorocarbons (CFCs), and they are thousands of times more powerful than carbon dioxide as a greenhouse gas. Because of this, most of them are regulated by Montreal Protocol (1989) and the majority of them are decreasing in concentrations around the world. Before their regulation, they were usually used as propellants and refrigerants.
